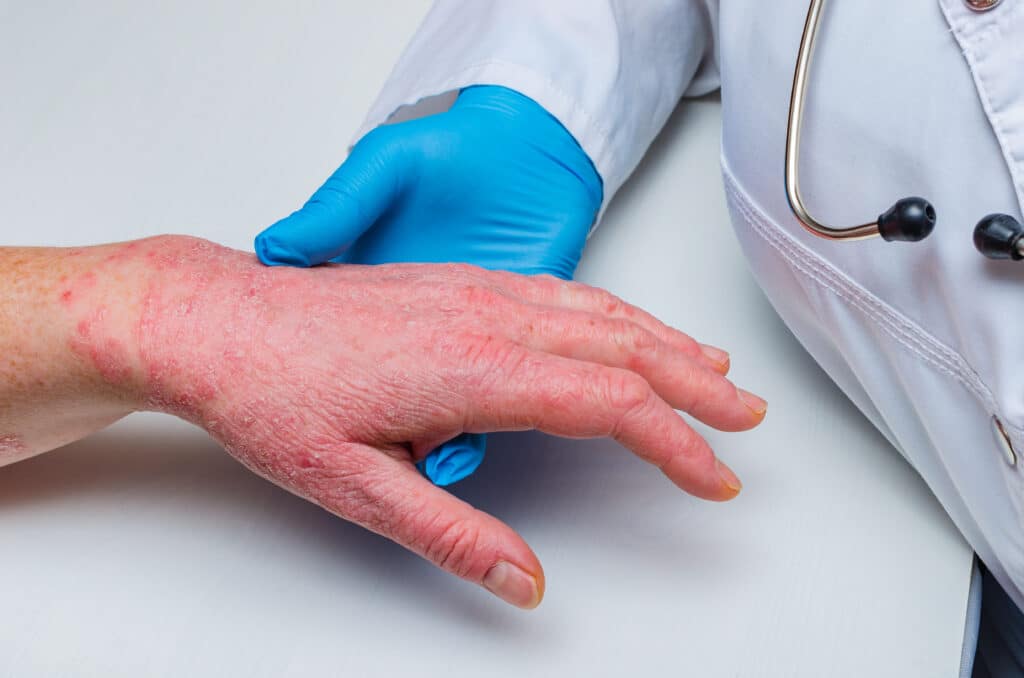
Results from a phase 2 study of gusacitinib, an oral Janus kinase and spleen tyrosine kinase inhibitor, demonstrated significant improvement in patients with chronic hand eczema (CHE).
Part A of this double-blind, placebo-controlled, multicenter study included 97 adult patients who were randomly assigned to receive either gusacitinib 40 mg, gusacitinib 80 mg, or placebo once a day for 12 weeks. In part B of the study, which continued through week 32, all patients received gusacitinib.
At week 16, the study showed that patients receiving 80 mg and 40 mg of gusacitinib experienced a decrease in modified Total Lesion Symptom Score (mTLSS) of 69.5% and 49% respectively, compared to 33.5% among placebo patients.
Furthermore, by week 16, 31.3% of those taking 80 mg and 21.2% of those taking 40 mg showed Physician Global Assessment (PGA) improvement compared to only 6.3% of the placebo group.
In looking at the hand eczema severity index (HECSI), the study also revealed that patients taking 80 mg of gusacitinib achieved a 73.3% decrease compared to placebo at 21.7%. Patients receiving 80 mg also experienced a significant decrease in hand pain.
A total of 33.3% of patients who received 40 mg, and 28.2% of those receiving 80 mg, had a marked improvement at week two in Patient Global Assessment compared with 0% in placebo. This continued to increase to week 16 with results showing 36.4% and 40.7% for 40 mg and 80 mg groups respectively compared to 18.8% for placebo.
According to the study, gusacitinib was well tolerated. The most common adverse events occurring in at least 10% of the patients were mild to moderate and included headaches, nausea, and short-term upper respiratory tract infections. These were most prevalent in those receiving 80 mg of gusacitinib.
The study concluded that gusacitinib showed rapid improvement in CHE patients and was well tolerated, and therefore warranted further investigations.
















